Table of Contents
Page created on March 5, 2018. Last updated on August 21, 2020 at 11:42
Learning objectives
- What are the functions of cholesterol?
- Describe the biosynthesis of cholesterol
- Which enzymes in cholesterol synthesis are head-to-head reactions, and which are head-to-tail reactions?
- Describe the function of the different lipoproteins
- Why are ketone bodies needed?
- Why does the liver have two forms of HMG-CoA synthase?
- Describe the biosynthesis of ketone bodies
- Describe the degradation of ketone bodies
Cholesterol
Cholesterol is a very important molecule. It’s a major component of cell membranes, which increases the fluidity and stability of the membrane. It’s also a precursor for vitamin D, bile acids, and steroid hormones.
Cholesterol is normally produced in the liver and transported to organs by lipoproteins like LDL. However, lipoproteins can’t penetrate the blood-brain barrier, so the brain synthesizes cholesterol on its own.
Synthesis:
Cholesterol synthesis takes place in the cytoplasm.
Synthesis begins with the thiolase reaction, a reversible reaction that combines two acetyl-CoA into one acetoacetyl-CoA. Then, acetoacetyl-CoA and acetyl-CoA are combined to yield HMG-CoA. The next step is the committing step; the reduction of HMG-CoA to mevalonate by HMG-CoA reductase. It is at this committing and irreversible step that regulation happens.
Mevalonate is then phosphorylated twice to yield the two isoprenes, isopentenyl-PP and dimethylallyl-PP. These combine to geranyl-PP, then to farnesyl-PP. Both reactions are head-to-tail condensations. Farnesyl-PP then goes together with another farnesyl-PP to give squalene in a head-to-head condensation reaction. Squalene then undergoes many reactions to give cholesterol.
| Reaction | Reaction type |
| First reaction of prenyl transferase | Head-to-tail condensation |
| Second reaction of prenyl transferase | Head-to-tail condensation |
| Squalene synthase | Head-to-head condensation |
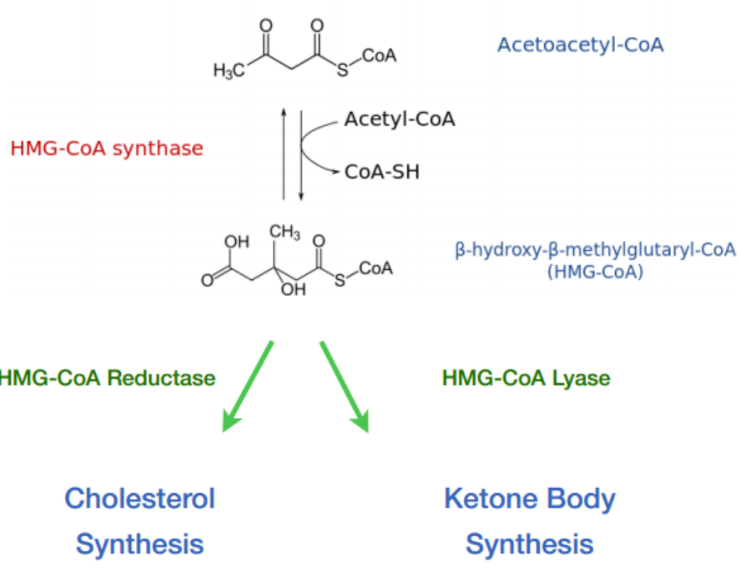
The ways HMG-CoA can take; either to cholesterol or to ketone bodies.
Cholesteryl esters
Cholesterol can be esterified by two enzymes, ACAT and LCAT.
ACAT is an intracellular enzyme which esterifies excess free cholesterol. Free cholesterol is toxic to cells and so to prevent toxicity excess free cholesterol is stored as cholesteryl ester.
LCAT is an extracellular enzyme found in blood. It esterifies cholesterol which is inside HDL.
Regulation:
Regulation is covered in topic 10.
Lipoproteins
Lipoproteins are complexes of protein, cholesterol, phospholipids, and fat. Their function is to transport fat and cholesterol around in the body. The protein part of lipoproteins is called apolipoprotein.
Different lipoproteins have different roles and compositions. Five types of lipoproteins exist. Chylomicrons, VLDL, IDL, LDL, and HDL.
Chylomicrons transport triacylglycerols from the intestines to the tissues of the body. They’re mostly comprised of triacylglycerols and contain very little cholesterol. After absorbing fat from food, intestinal epithelial cells secrete chylomicrons into the lymphatics, which eventually drain into the circulation. Inside the circulation lipoprotein lipase cleaves off free fatty acids from the chylomicrons. Peripheral tissues take in these free fatty acids.
When the chylomicron has been depleted of triacylglycerols it’s known as a chylomicron remnant. The liver will take up the chylomicron remnant.
VLDL (very low-density lipoprotein) transports triacylglycerols from the liver to the tissues. It’s comprised mostly of triacylglycerols. In the circulation lipoprotein lipase will cleave free fatty acids from VLDL, which peripheral tissues can take in.
After VLDL has given off much of its triacylglycerols it has become IDL (intermediate-density lipoprotein). The liver will convert IDL to LDL with the help of hepatic lipase.
LDL (low-density lipoprotein) is a remnant of IDL, which is a remnant of VLDL. LDL is comprised of mostly cholesterol; it contains few triacylglycerols. Peripheral tissues which need cholesterol will express an LDL receptor. When LDL binds to this receptor the LDL will be endocytosed by the cell, providing the cell cholesterol.
HDL (high-density lipoprotein) is involved in the reverse cholesterol transport, transporting excess cholesterol from peripheral cells back to the the liver. HDL precursors are formed by liver and intestine. Peripheral tissues which need to get rid of cholesterol will take in the HDL precursors, pack them with cholesterol, and secrete them back to the liver. The liver can then excrete excess cholesterol as bile.
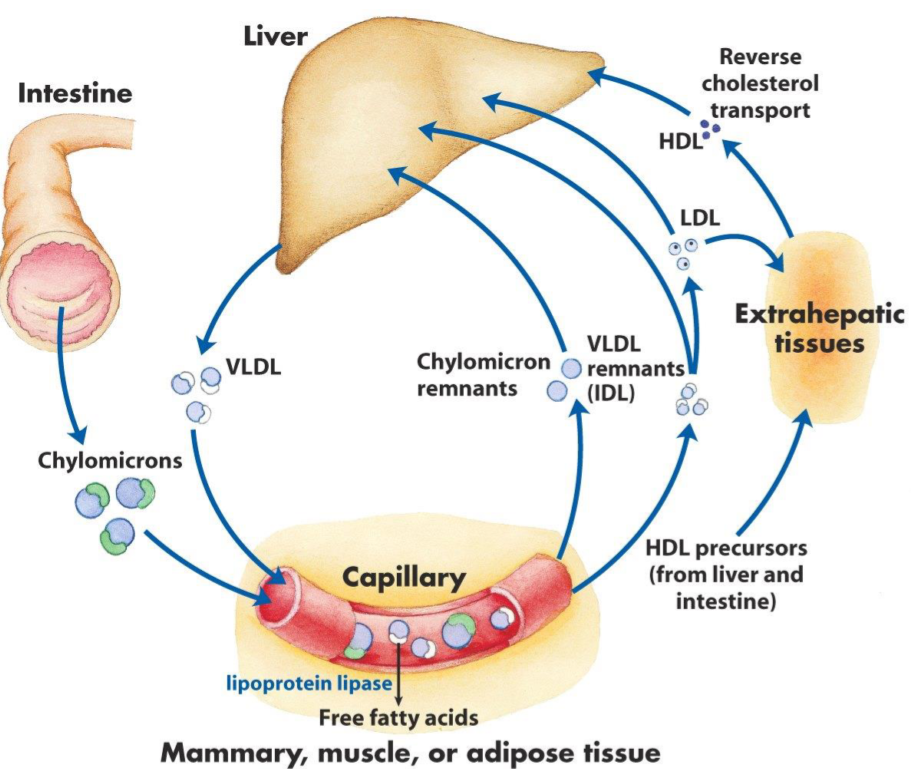
How lipoproteins are transported in the body. Note that extrahepatic tissues just mean tissues that are not the liver, like muscle and adipose tissue.
Ketone bodies
Ketone bodies are “backup” energy molecules. In periods of fasting the blood sugar level will drop. The body can break down proteins to glucogenic amino acids, which can be converted into glucose. This prevents the blood glucose level from dropping to zero, which would be fatal. However, fatty acids cannot be converted into glucose. So how does your body utilize its fat stores during starvation?
The liver can’t convert fatty acids to glucose, but it can convert them to ketone bodies. Ketone bodies are an alternative energy source which all tissues can utilize. These ketone bodies are produced by the liver and will be transported in the blood to other tissues and then be broken down into acetyl-CoA to yield energy to fuel the TCA cycle, in the same way glucose would.
Fatty acids cannot cross the blood-brain barrier, but ketone bodies can. The brain is therefore especially dependant on ketone bodies, as they can be used both as energy and to synthesise fatty acids in the brain.
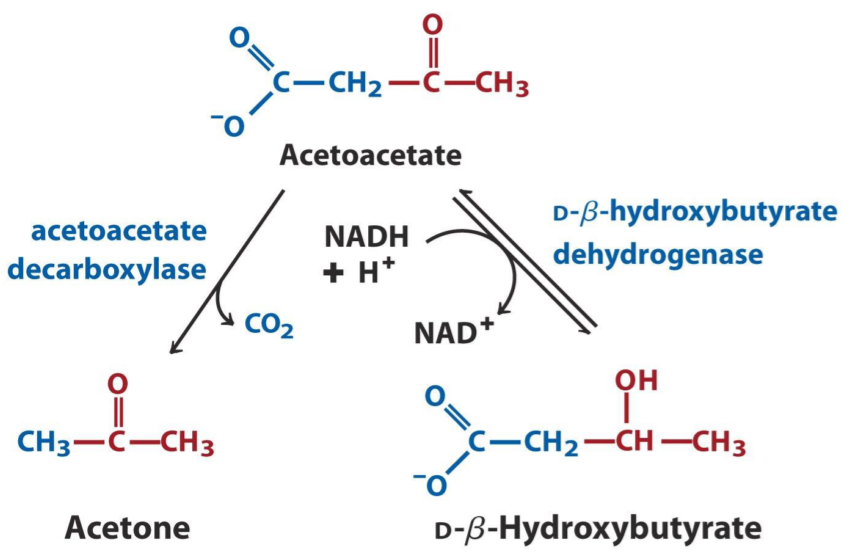
There are three ketone bodies, acetoacetate, β-hydroxybutyrate, and acetone.
How ketone bodies are converted into each other. All three of these are ketone bodies. Note that D-β-hydroxybutyrate isn’t strictly a ketone, but is still considered a ketone body.
Synthesis
Ketone bodies are only produced in the mitochondria of the liver cells. It uses acetyl-CoA yielded from beta-oxidation (which also occurs in the mitochondria). This means that citrate plays no role in ketone body synthesis, because there is no need to transport acetyl-CoA across the mitochondrial membrane.
Just like cholesterol synthesis, ketone body synthesis begins with the thiolase reaction, a reversible reaction that combines two acetyl-CoA into one acetoacetyl-CoA. Then, acetoacetyl-CoA and acetyl-CoA are combined by HMG-CoA synthase to yield HMG-CoA. This HMG-CoA synthase is mitochondrial.
An exclusive enzyme called HMG-CoA lyase breaks down HMG-CoA into acetoacetate and acetyl-CoA.
When the body has synthesized acetoacetate, it can use this to create the other two ketone bodies. Acetone isn’t very important though; the other two are much more important in the body.
Breakdown
During the breakdown of ketone bodies, D-β-hydroxybutyrate is oxidized to acetoacetate, which receives a CoA from succinyl-CoA in the β-ketoacyl-CoA transferase reaction. Acetoacetyl-CoA is then broken down into two acetyl-CoA by thiolase.
Summary
- What are the functions of cholesterol?
- Cholesterol provides stability and fluidity to cell membranes
- Cholesterol is a precursor of many hormones
- Describe the biosynthesis of cholesterol
- Occurs in the cytosol of hepatocytes
- Describe the reactions
- Which enzymes in cholesterol synthesis are head-to-head reactions, and which are head-to-tail reactions?
- The two reactions of prenyl transferase are head-to-tail reactions
- The reaction of squalene synthase is a head-to-head reaction
- Describe the function of the different lipoproteins
- Chylomicron transports dietary fats to peripheral tissues
- VLDL transports hepatic fats to peripheral tissues
- IDL is a remnant of VLDL. It transports fats and cholesterol to the liver
- LDL is a remnant of IDL. It transports hepatic cholesterol to peripheral tissues
- HDL transports peripheral cholesterol to the liver.
- Why are ketone bodies needed?
- Because fatty acids can’t be used for gluconeogenesis. The only way for fatty acids to provide energy for other tissues is to convert them to ketone bodies
- Why does the liver have two forms of HMG-CoA synthase?
- The cytoplasmic form is for cholesterol synthesis
- The mitochondrial form is for ketone body synthesis
- Describe the biosynthesis of ketone bodies
- Occurs in the mitochondria of hepatocytes
- Describe the reactions
- Describe the degradation of ketone bodies
- Describe the reactions
Hey Nikolas,
where did you find the schemtaic depiction of the transport system of the lipoproteins? Could you cite your source?
Thank you.
I took the figure from our lecture, but they must’ve taken it from some book. I tried to figure out which book. It’s not Lehninger or “Basic Medical Biochemistry: A clinical approach”.
I think it might be from the book “kaplan mcat biochemistry”, but I’m not sure.