Page created on November 23, 2018. Last updated on December 18, 2024 at 16:56
We’re getting into biochemistry territory
I recommend starting with the relevant medical biochemistry topic before getting into this.
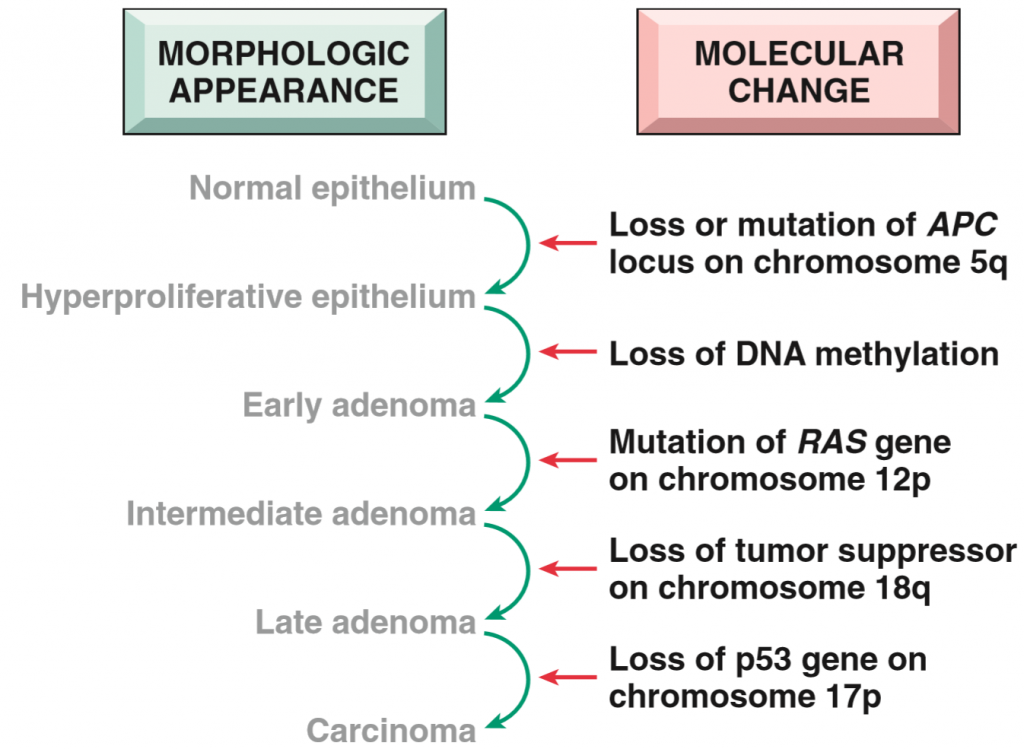
All cancers start with some genetic damage. This genetic damage is always nonlethal, meaning that the damage isn’t big enough to actually kill the cell, however it is definitely big enough to cause some serious damage. The process where cancer develops is called carcinogenesis.
Nonlethal genetic damage causes mutations and can be due to many factors. Common factors include environmental agents like chemicals, radiation and viruses.
Take a step back and think about this: All tumors begin as a single progenitor cell that acquires some genetic damage, which then undergoes clonal expansion, where this cell divides into two, and these two divide into four, and so on. All tumors originate from one single cell, meaning that every tumor is monoclonal.
Five types of genes are normally mutated in cancers:
- Proto-oncogenes
- Tumor suppressors
- Genes that regulate apoptosis
- Genes involved in DNA repair
- Micro RNAs
Let’s talk about each of them in detail.
Gene mutations in cancer
Some proteins have different names in biochemistry and in pathology. What you might now as p53 is here called TP53, and pRb is here called RB.
Proto-oncogenes are genes that, when not mutated, have some growth-promoting effect on the cell. They often code for transcription factors, growth factors, growth factor receptors, signal transmission, cell cycle regulators or proteins involved in cell survival. Characteristic for proto-oncogenes is that they’re dominant, meaning that only one allele (copy of the gene) needs to be mutated for the proto-oncogene to become an oncogene. The gene product of an oncogene is called an oncoprotein. These oncoproteins usually lack regulatory elements.
A classic example of a proto-oncogene is one that codes for a growth factor receptor. When growth factor receptors are activated, by binding a growth factor in the blood, will they signal to the cell that it should proliferate. However, if the proto-oncogene is mutated and produces an oncogene instead can the growth factor receptor be constantly activated, despite there being no growth factors in the blood! This initiates uncontrolled cell division of the cell.
Tumor suppressors are genes that, when not mutated, prevent uncontrolled growth. They can be considered “brakes” on cell growth. When these genes are mutated, their regulatory function is lost, and the cell is less protected against uncontrolled growth. Tumor suppressors are considered to be recessive, meaning that both alleles must be mutated for the function to be lost. This implies that loss of tumor suppressor function is less frequent than oncogene formation. Recent research however has shown that even loss of a single tumor suppressor allele can promote cancer transformation.
Tumor suppressors are divided into two groups, governors and guardians. Governors are the “classic” tumor suppressors that function as brakes on cell proliferation. RB belongs to this group. Guardians are genes that are responsible for sensing damage on the genome, like TP53, which has earned the nickname guardian of the genome for its importance.
Loss (of function) of the guardian genes don’t directly cause cells to start proliferating like crazy, however a cell without a guardian accumulates mutations in proto-oncogenes and other tumor suppressors more easily.
Genes that regulate apoptosis or DNA repair don’t transform the cell directly either, but, like guardian genes, can be a factor in accelerating the accumulation of other mutations.
Recall from Biochemistry 1 that microRNAs (miRNAs) are small non-coding RNA molecules that are inhibitory. Some cancers are associated with dysregulation of certain miRNAs. An miRNA that is involved in cancers called an oncomir.
There are 7 fundamental changes in a cell that causes it to acquire a malignant phenotype. They are:
- Self-sufficiency in growth signals
- Insensitivity to anti-growth signals
- Escape from apoptosis
- Limitless replicative potential
- Sustained angiogenesis
- Ability to invade and form metastasis
- Inability to repair DNA damage
Growth factors and growth factor receptors
The most important growth factor receptors in cancer development are the tyrosine kinase receptors. Recall from Biochemistry 1 that these receptors bind a ligand on the outside, which triggers the tyrosine kinase activity of the receptor on the inside of the cell. The receptor will then phosphorylate tyrosine residues on a substrate which mediates the signal further.
Oncogenic versions of tyrosine kinase growth factor receptors have their tyrosine kinase activity constitutively activated, meaning that it’s always active, even without binding the ligand. This causes the cell to continuously “receive” the stimulus to proliferate.
Oncogenic growth factor receptors aren’t necessarily just the result of gene mutation, they can form in gene rearrangement or overexpression of the oncogene as well.
Now’s a good time to explain the difference between gene overexpression and gene amplification. Amplification occurs when the gene itself is copied in the genome, which can cause hundreds of the same gene in the genome. Overexpression occurs when a single gene is overexpressed, due to increased upregulation of that gene.
Here are some examples of some important mutations and translocations in oncogenes with their corresponding tumor type:
| Oncogene | Physiological function | Genetic alteration | Tumor |
| RET | Receptor for Glial cell line-Derived Neurotropic Factor (GDNF) | Germ-line point mutation | Multiple endocrine neoplasia type 2 (Medullary thyroid cancer + pheochromocytoma + parathyroid adenoma) |
| RET | Somatic point mutation | Medullary thyroid cancer | |
| FLT | Not important | Point mutation | Myeloid leukaemia |
| PDGFRβ and ETS | Growth factor receptor and transcription factor | Fusion of the two genes. t(5;12) | Chronic myelomonocytic leukaemia |
| KIT or PDGFRα | Growth factor receptor | Mutation | 85% of all gastrointestinal stromal tumors (GIST) |
A germ-line mutation is a mutation was present in the germ cell, i.e. it’s not a spontaneous mutation in one cell, it’s a mutation all cells in that body has, that has been inherited from the parents. Somatic mutations are mutations that occur in somatic cells, where only that somatic cell (and all its descendants) carry the same mutation.
One of these entries warrants more explanation. The fourth line shows how translocation of a gene into another gene can cause cancer. In this case is the gene for PDGFRβ (platelet-deriver growth factor receptor beta), which is on chromosome 5, is translocated into the gene for ETS, which is on chromosome 12. The denotation t(5;12) indicates a gene translocation from chromosome 5 to chromosome 12.
Imatinib and sunitinib are drugs that can be used to treat gastrointestinal stromal tumors.
While mutation and translocation can cause oncogene formation is overexpression more common than the other two. There are two good examples for this:
ERBB1 is a gene for another growth factor receptor. The growth factor in this case is epidermal growth factor (EGF). ERBB1 is overexpressed in:
- 80% of lung squamous cell carcinomas
- >50% of glioblastomas
- 80-100% of head and neck carcinomas
ERBB2, also called HER2, is another tyrosine kinase receptor, however we still don’t know its physiological ligand. HER2 is overexpressed in:
- 15-25% of breast carcinomas
- Adenocarcinomas of ovary, lung, stomach and salivary glands
Biological treatment for HER2 positive breast carcinoma is a monoclonal antibody called herceptine. These antibodies cause the breast carcinoma cells to internalize the HER2 receptor, limiting its harmful potential and effectively treating the cancer.
Hey there 🙂
Thanks for putting this together. Just a minor correction on the Multiple Endocrine Neoplasia 2. It should be medullary thyroid cancer, not thymic.
Fixed. Thank you for the correction and glad I could help!
Hi
The somatic point mutation part of the table still shows “thymic” instead of “thyroid”
Not anymore it doesn’t