Table of Contents
Page created on March 19, 2018. Last updated on December 18, 2024 at 16:56
Seeing
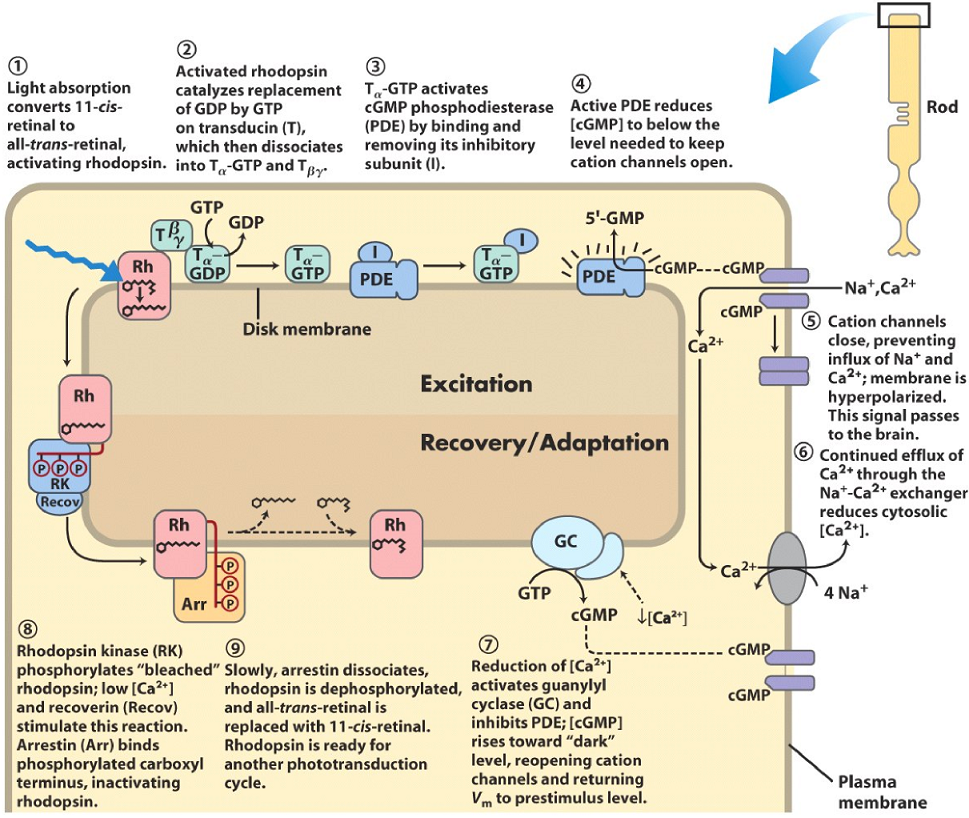
Rods, the cells that sense light, are unique in one way compared to other cells of the nervous system. While other cells are depolarized when they become activated, rods are hyperpolarized! When resting, the rods are depolarized. This happens because in most cells, a signal is propagated when the amount of a second messenger increases, in rods this happens when [cGMP] decreases, as we will see.
The rod cell membrane contains multiple ion-channels that are important here. The most important is the cGMP-gated cation channel (in purple and on the right on the figure) which allows sodium and calcium ions to enter the cell. This inward flow of cations is called the “dark current”, as it flows only when the rod is not stimulated by light. In the dark, the levels of cGMP in the cell is high, which allows the cGMP-gated cation channel to stay open and the dark current to flow.
Also attached to the membrane is a protein called rhodopsin (red on the figure). This protein is a G-protein coupled receptor, and consists of two parts, an opsin, and a 11-cis retinal molecule. The G-protein that is coupled to rhodopsin is called transducin (green on the figure).
When a photon meets the rhodopsin protein, it will convert the 11-cis retinal to all-trans retinal. The rhodopsin protein is now activated, which activates tranducin, which will activate membrane-bound phosphodiesterase (PDE, blue on the figure). The rhodopsin with all-trans retinal is now “bleached”, because it loses its color.
Phosphodiesterase will cleave cGMP to GMP which will decrease the [cGMP] in the cell. Following this, the [cGMP] levels drop so low that the cGMP-gated cation channel will close and no longer allow cations like Ca2+ and Na+ to flow into the cell. This hyperpolarizes the membrane, which propagates the signal to the brain.
Now that the cGMP-gated cation channel is closed, and a Na+-Ca2+ exchanger (grey on the figure) will pump Ca2+ ions out, the levels of [Ca2+] in the cell will drop. After the levels of [Ca2+] reach around 100 nM, guanylyl cyclase will be activated and PDE inhibited, which will cause cGMP levels to increase back to the levels before the photon arrived. This causes the cGMP-gated cation channel to open again.
After [Ca2+] dropped when cGMP-gated cation channels were closed, a protein called rhodopsin kinase will phosphorylate rhodopsin. This action is stimulated by low [Ca2+] and a protein called recoverin. Another protein called arrestin (Arr) will bind to phosphorylated rhodopsin, which inactivates it.
Slowly and after a period of time, arrestin will dissociate, rhodopsin will be dephosphorylated and the all-trans retinal is replaced with a 11-cis-retinal. The cell is now ready to be stimulated again.
The phosphorylation of rhodopsin is important in adapting the rods to different levels of ambient light. When there is a lot of ambient (background) light, like outside on a sunny day, rhodopsin will be heavily phosphorylated. The phosphorylation makes rhodopsin harder to activate, so that the background light doesn’t blind you. In a dark room, rhodopsin will be completely dephosphorylated, so that it’s more sensitive to small amounts of light.
While talking about the eye, it’s fitting to mention the disease where the lens, which is usually transparent, becomes opaque. This disease is called cataract, and can be caused by errors in galactose metabolism, diabetes, old age or other factors.
Smelling
The olfactory receptors recognize the smell of molecules based on their shapes. Two molecules with similar shapes will bind to the same olfactory receptor, which will send the same signal to the brain. Because of this, the olfactory epithelium must consist of many receptors that each recognize a shape and not a specific molecule. This means that chirality is important in the smell of a molecule. For example, R-carvone smells like spearmint, but its enantiomer S-carvone smells like caraway. Each olfactory neuron expresses only one kind of receptor.
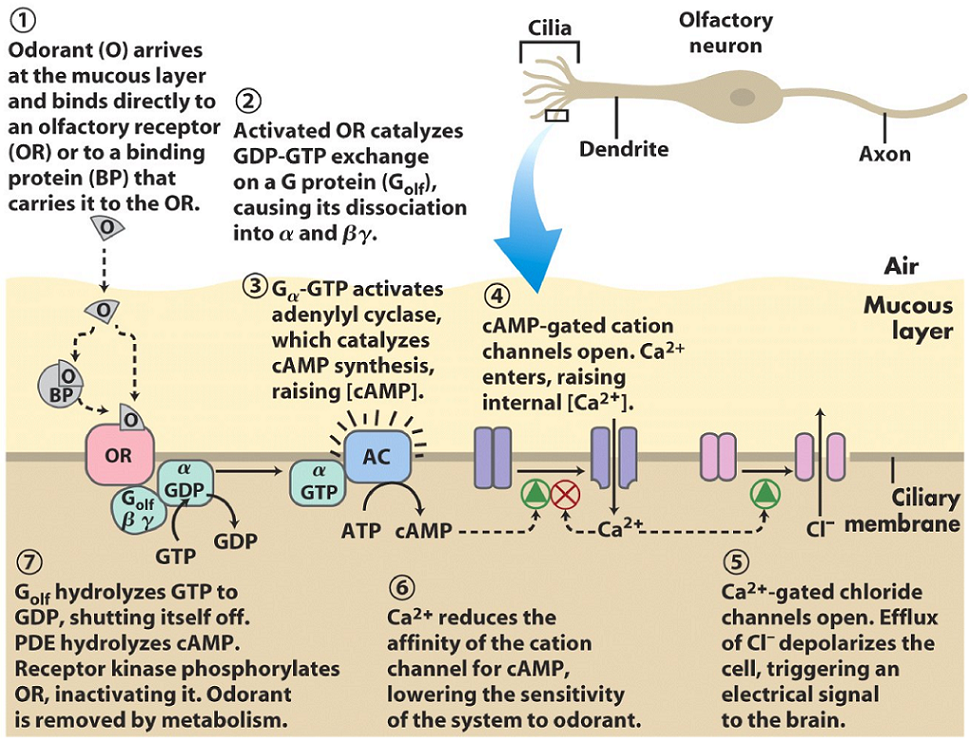
The olfactory receptor is situated in the ciliary membrane of the cilium of an olfactory neuron. It can bind the odorant (the molecule to be smelled) either directly, or by the function of a binding protein, that first binds to the odorant and then to the receptor. The receptor is a G-protein coupled receptor, which will activate membrane-bound adenylyl cyclase. This enzyme will convert ATP to cAMP.
There are two ion-channels in the ciliary membrane. The first is a cAMP-gated cation channel, which will open when the [cAMP] increases in the cell. After opening, Ca2+ will flow into the cell. Ca2+ also inhibits the effect of cAMP on the channel as a negative feedback mechanism.
The second ion channel is a Ca2+-gated chloride channel. When [Ca2+] increases due to the other ion channel, this ion channel will open, which allows Cl– to escape the cell. This Cl– efflux depolarizes the cell, which propagates the signal to the brain.
After the signal has been sent to the brain, it’s time to reset the cell so that it’s ready for the next odorant. The increasing [Ca2+] levels in the cell will continue to close the cAMP-gated cation channel, which reduces the signal. Phosphodiesterase will hydrolyse cAMP to AMP. The G-protein will hydrolyse GTP to GDP, which inactivates it. A receptor kinase will phosphorylate the olfactory receptor, which inhibits it. Finally, the cell is ready for a new odorant.
Tasting
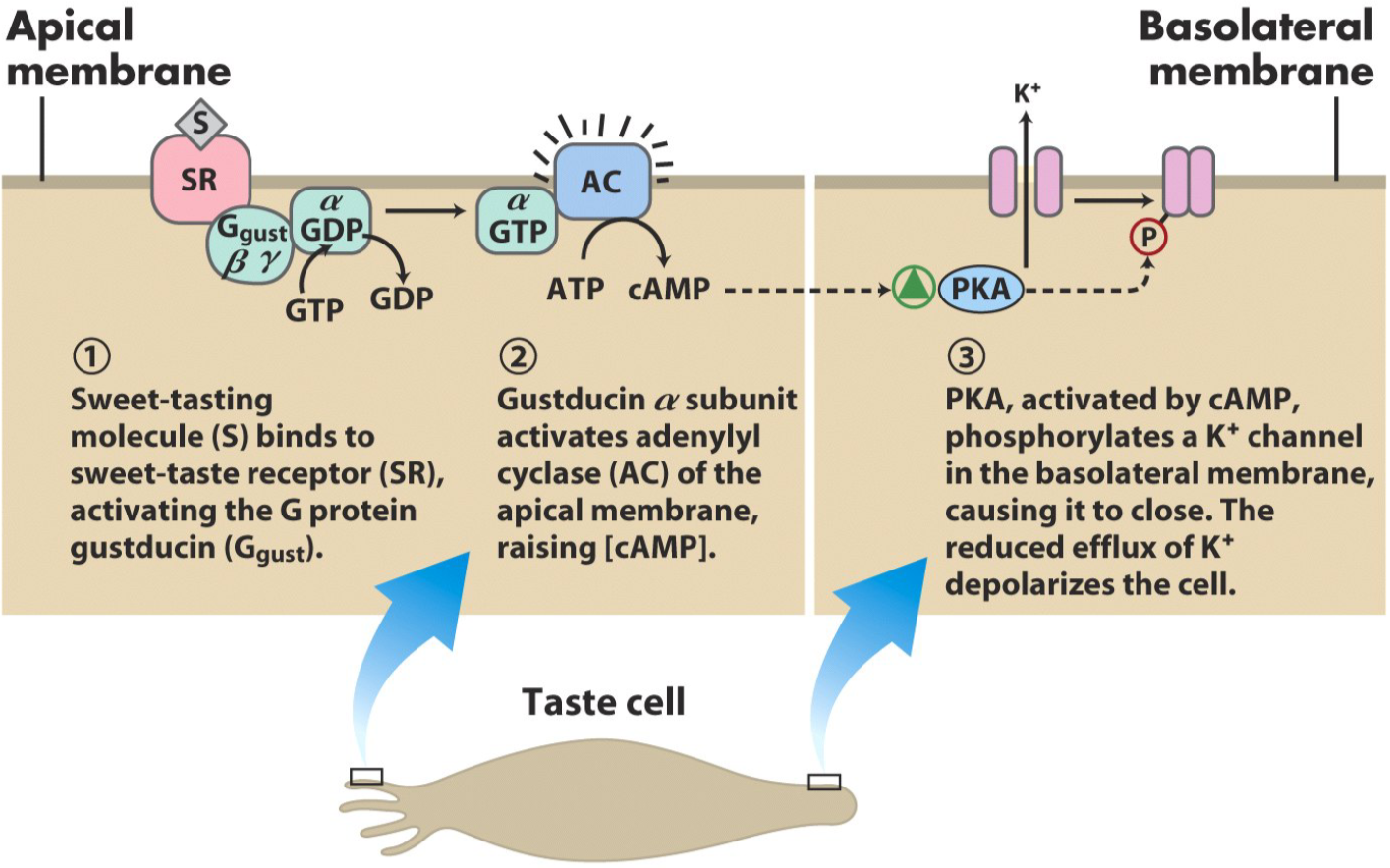
The process of tasting something sweet is similar to the process of olfaction, but simpler. The sweet-tasting molecule binds to the sweet-taste receptor, another G-protein coupled receptor. The G protein the receptor is coupled with in this case is called gustducin. Gustducin will activate adenylyl cyclase, which will increase intracellular [cAMP]. This will activate PKA, which will phosphorylate a K+ channel on the basolateral membrane, which will cause it to close. The reduced outflow (efflux) of K+ depolarizes the cell, which propagates the signal to the brain.
The tasting of umami and bitter is also mediated by G-proteins, but their mechanism is not be known. Sour taste is mediated by proton pumps, which pump H+ ions from the food into the cell. Salty taste is mediated by a special sodium channel called amiloride-sensitive sodium channel, which is also found in the distant tubule of the kidney. Amiloride is a medication used to close this type of channel, to increase diuresis in the kidney, while having the (theoretical) side effect that food doesn’t taste as salty.
Hot food is sensed by the vanilloid receptor, which binds capsaicin, a compound found in hot peppers and the like. The receptor activates a hot, burning sensation.
Hearing
Hearing doesn’t depend on any complicated biochemical mechanism. It’s more of a physiology thing actually. Hair-cells in the ear are surrounded by a fluid called the endolymph, which moves when sound waves enter the ear. The hair cells have many stereocilia. Two adjacent stereocilia are bound at the tip by a protein called the tip link. This protein can detect changes in its own length, which will induce changes in the cell that releases neurotransmitters from the basal part of the cell, which reaches a nerve cell that will propagate the signal to the brain.
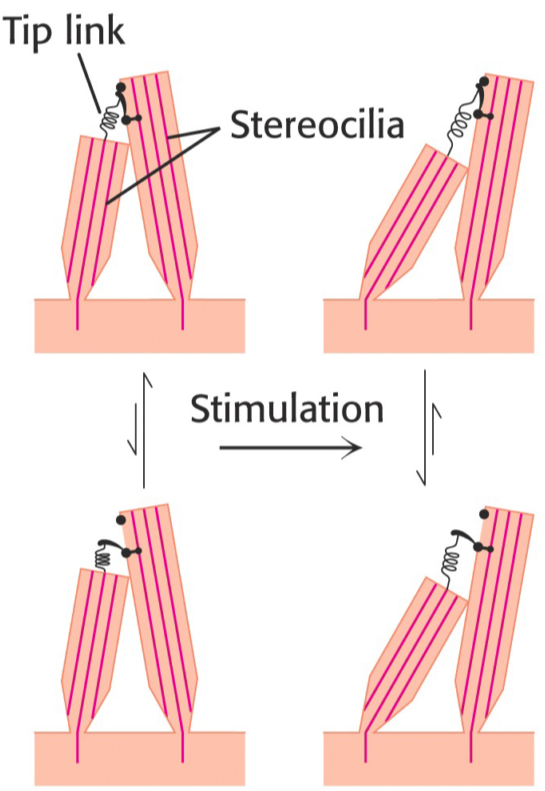
For hearing, I feel like there is a lot of biochemistry here which can be mentioned. In endolymph the potential is: +80 mV and in the hair cell it is –60 mV –> Making a +150 mV gradient. The longest cilia on the Hair cell is the Kinocilia and when the Stereocilia are bent towards the Kinocilia they open Mechanosensitive K+ channels. Big Membrane potential as previously mentioned would make K+ flow in very rapidly into the hair cell activating Voltage gated Ca2+ channels. These channels activate exocytosis of Glutamate which will bind to NMDA receptors (Ligand gated Na+ receptors) on the corresponding nerve of the area, creating an AP.
These topics follow the lectures, and the lectures don’t go into that detail. Ergo, it’s likely unnecessary to include.
It says above under the tasting section that Amiloride is used to block the sodium channel and decrease diuresis. wouldn’t blocking the sodium channel increase diuresis?
Good catch, it’s actually used as a diuretic.
“The process of seeing light. It sucks.”
Actually, I think it’s pretty cool. Your notes are really good, but why make people want to look away by saying “it sucks”? Why not inspire others to see the incredible small mechanisms and make others understand how amazing it is??
Every physiological and biochemical process in the body is cool. But some of them still suck. I’m glad you like it though.