Page created on April 4, 2018. Last updated on November 19, 2018 at 17:16
Affinity maturation
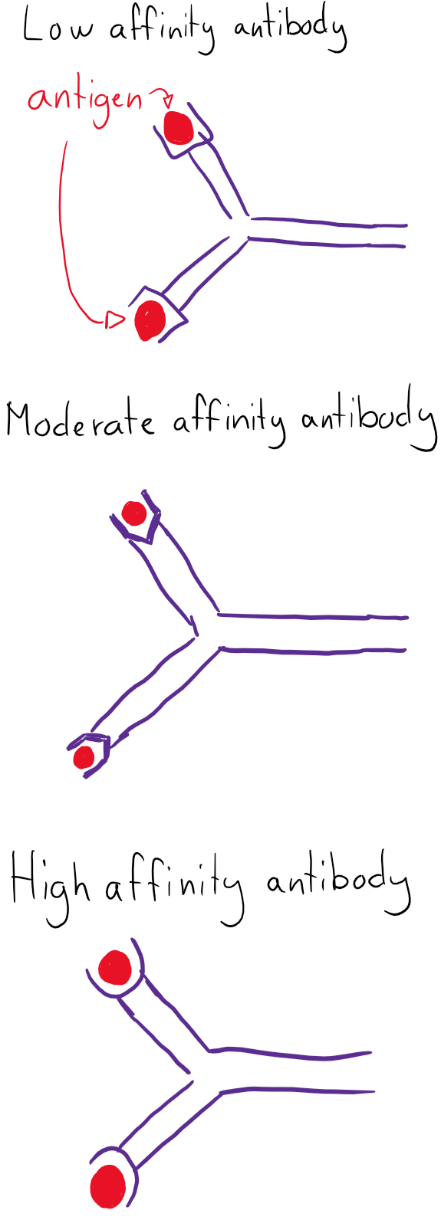
As written in the last topic, the follicular B-cells in the germinal center undergo a process called affinity maturation. This causes the B-cells to produce antibodies with higher affinity to the antigen than originally.
During the division of the centroblasts, an enzyme called activation-induced cytidine deaminase, or AID, which is activated by CD40 binding, will deaminate thymidine bases in the genes for the CDR regions to uracil. Because uracil is not meant to be present in DNA (only in RNA), they will be removed by enzymes, which will cause both DNA strands to break at many points in these genes. This DNA damage will be repaired by error-prone DNA-polymerases, which causes the mutations to appear in these genes.
While most of these mutations will create genes that code for dysfunctional or lower affinity antibodies, some are bound to yield antibodies with higher affinity than originally. The B-cells with lower affinity are destined for apoptosis, while B-cells with higher affinity are saved from it. This is called germinal center selection. From the original 1 B-cell that was activated by the TFH, approximately 100 000 clones are made, all of which undergo affinity maturation. After the selection, approximately 100 high-affinity B-cells remain.
In order to perform this selection, the centrocytes’ (B-cell’s) antibody’s affinity must be tested. Recall that the follicular dendritic cells store the antigen. The centrocytes’ antibody will try to bind to the stored antigen. If it binds with high affinity, it will be saved from apoptosis.
Isotype switching
Isotype switching is the process of how the B-cells decides what type immunoglobulin isotype it is going to produce, like IgG, IgA or IgE. This also happens at the gene-level but is not random like affinity maturation is. Recall that the isotype of the immunoglobulin depends on the constant domain of the heavy chain of the immunoglobulin.
Every B-cell has the genes for every heavy chain isotype. The genes for the different immunoglobulin heavy chain isotypes are separated by so-called switch regions. In a fashion similar to VDJ recombination, two of these switch regions will attract each other, which creates a loop in the DNA. This loop contains the genes for the heavy chain isotype that will not be expressed and is then cleaved away. What remains are the untouched VDJ segments, and the gene for the constant domain that ends up closest to the VDJ segment will be expressed. The AID enzyme is important in cutting out the unused constant domain genes. The figure below explains the process better.
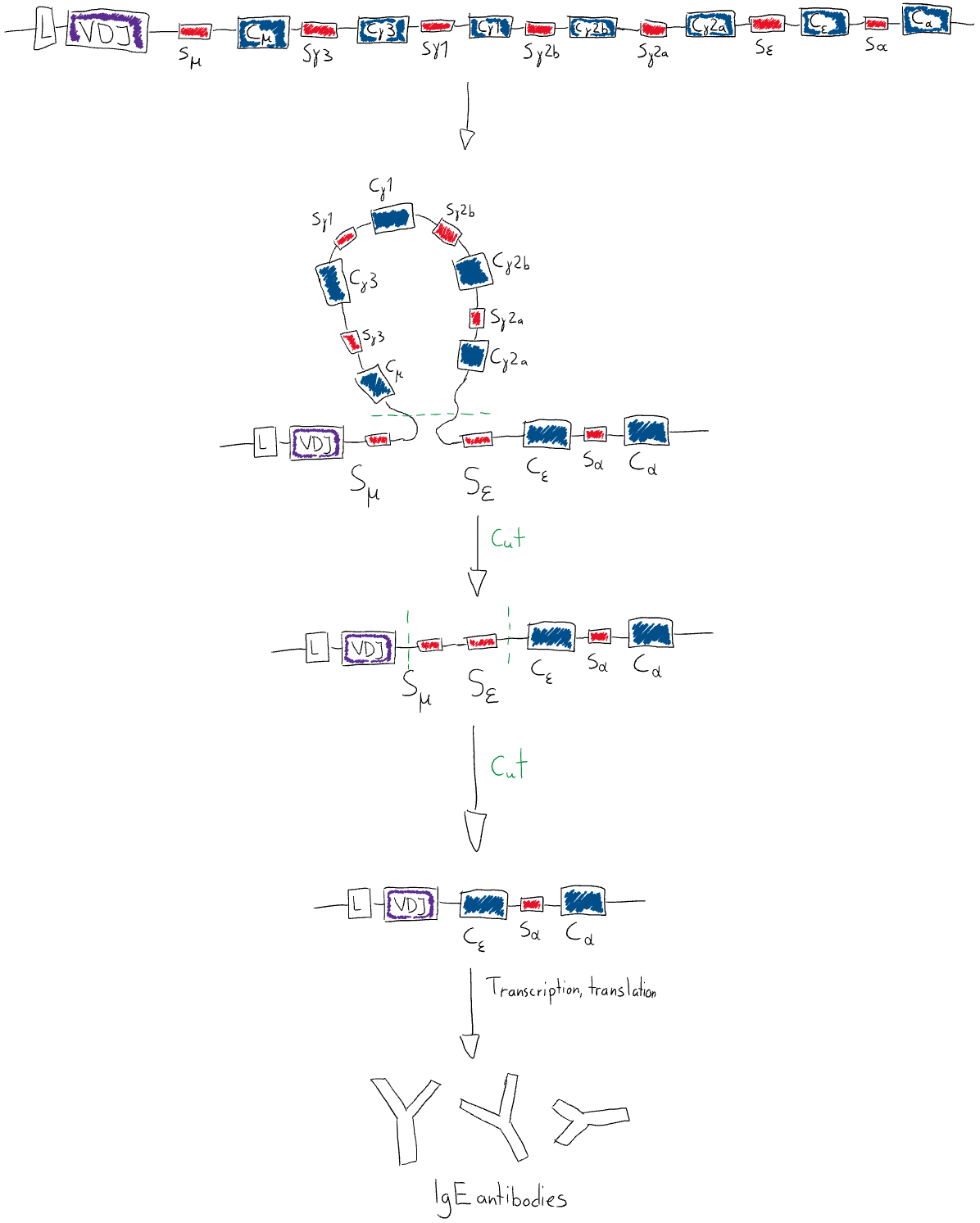
What decides what isotype of immunoglobulins a B-cell should “switch” to? Cytokines of course. Depending on the type of pathogen and its entry into the body, and whether there is an inflammation or not, different cytokines will be produces by the body. For example, when there is a parasitic infection, IL-4 will be produced, which will cause B-cell to isotype switching to IgE and T helper cells to differentiate into TH2 cells. IFN-γ will induce B-cell isotype switching to IgG.
After this
The high affinity centrocytes will eventually enter the circulation to become plasmablasts, where they continue to produce antibodies. After the infection, they migrate to the bone marrow where they become plasma cells, that will continue to produce high affinity antibodies for years. The centrocytes destined to become plasmablasts have a high amount of a protein called Blimp-1.
Some of the high affinity centrocytes will not secrete antibodies but become memory B-cells. They will circulate in the blood and reside in mucosa. Here they can survive for years and renew themselves after that. This enables the memory from the germinal center reaction to last for a lifetime.