Table of Contents
Page created on November 22, 2018. Last updated on December 13, 2018 at 14:48
Characteristics of benign and malignant tumors
Benign and malignant tumors are different in four important aspects. They’re best summed up in a table:
| Characteristic | Benign tumors | Malignant tumors |
| Differentiation/anaplasia | Well differentiated. Cells resemble normal cells. | Variable differentiation, from well-differentiated to undifferentiated (anaplastic) cells. |
| Rate of growth | Slow and progressive. Mitotic figures are rare and normal. | Erratic, can be slow or fast. Mitotic figures are numerous and can be abnormal |
| Local invasion | Expansile border, well-circumscribed tumor. Tumor often has capsule | Hard to circumscribe. Infiltrative border. Usually no capsule. |
| Metastasis | Never | Frequently |
Differentiation
Differentiation is a term used to describe how much the cells of the tumor resembles the equivalent, healthy cell type. This includes both morphology and function, for example well-differentiated hepatocyte carcinomas secrete bile, while a poorly differentiated one wouldn’t secrete bile. Well-differentiated squamous cell carcinomas produce keratin, while poorly differentiated ones won’t. Well-differentiated endocrine tumors produce hormones, and so on. In fact, the hormones produced by some tumors can give physicians a sign that something is wrong based on blood tests.
Benign tumors are always well-differentiated, while malignant tumors can be anywhere from well-differentiated to completely undifferentiated (anaplasia). Differentiation isn’t a binary thing (either well or poorly differentiated); it’s a spectrum. Any cancer can be anywhere from well to poorly differentiated. The more poorly a tumor is differentiated, the more aggressive it is, and the worse the prognosis. Several techniques can be used to determine the degree of differentiation, like immunohistochemistry (IHC) and in-situ hybridization (ISH).
The degree of differentiation of cancers is important in their grading, which we’ll look at in topic 50.
Rate of growth
Most benign tumors grow slowly, while most cancers grow much faster. Cancers can spread, both locally and systemically (metastasizing), to eventually cause death. There are exceptions to this rule; some benign tumors grow faster than cancers. The rate of growth in cancers usually correlates inversely with their degree of differentiation, meaning that poorly differentiated cancers tend to grow the fastest.
The growth pattern of cancers can vary. They don’t necessarily grow steadily; a cancer could grow slowly for many years until one of the cancer cells acquire a new mutation that causes it and the tumor to grow like crazy. Research has shown that most cancers take years or even decades to produce symptoms and become clinically relevant.
If cancers grow quickly enough will the tumor blood supply often be insufficient to supply the central areas of the tumor, creating a necrotic “core” inside the tumor.
The rate of tumor growth depends on three factors:
- The time it takes for the tumor cells to double in number, the doubling time of the tumor cells
- The fraction of the tumor cells that are actively proliferating, the proliferative fraction of the tumor
- How quickly tumor cells die, the death rate of the tumor cells.
Doubling time of tumors differ between tumors, but the biggest determinant for how fast cancers grow is the proliferative fraction. Not all tumor cells in a tumor is currently replicating, only a fraction of them. This fraction changes considerably in the development of the tumor. In the very early stages of cancer are the majority of cells proliferative, but this fraction decreases when the tumor grows. At the time where cancers become clinically detectable are only approx. 20% of the tumor cells proliferating.
Chemotherapeutic drugs act on proliferating cancers cells, meaning that the proliferative fraction of the tumor is very important for whether these drugs will have an effect or not. Neuroblastoma and Burkitt lymphoma are both cancers with very high proliferative fractions, and both responds relatively well to chemotherapy.
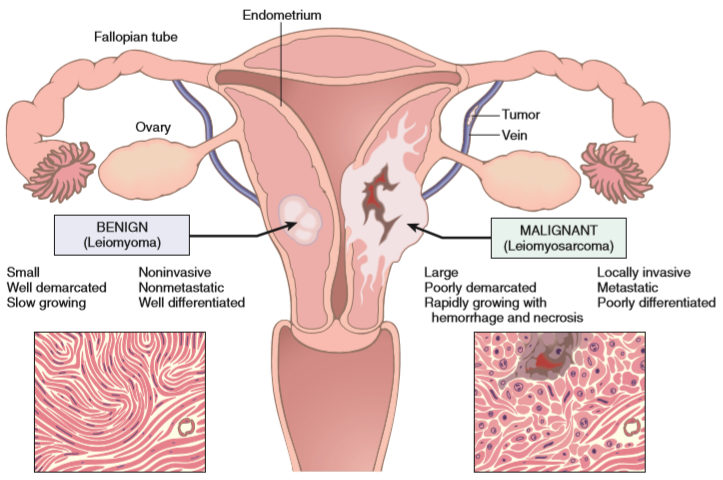
Local invasion
Benign and malignant tumors differ considerably in how they “treat” the tissue they grow in.
Benign tumors have a well circumscribed, expansile border, meaning that they don’t grow “into” the surrounding tissue but instead pushes it away as they expand. They have no ability to infiltrate, invade or metastasize to other tissues. They often form capsules, which separates them from the surrounding tissue. These properties mean that surgical removal is easy.
Malignant tumors are not well circumscribed, and they have an infiltrative border instead of an expansile one. Malignant tumors don’t push away the surrounding tissues but instead grow into them. They cross borders like basement membranes, enter vessels and penetrates whole walls of organs like the colon, uterus and bladder. They can even break through to the surface of the skin, as seen in the breast carcinoma preparation.
You can compare macropreparations 35 and 36 to see the difference between benign and malignant tumors. Histopathology slide 36 shows an infiltrative border.
Metastasis
Metastases (plural of metastasis) are secondary implants of a tumor that aren’t continuous with the primary tumor, and that are located in remote tissues. Approximately 30% of newly discovered tumors have already produced clinically relevant metastases. An additional 20% have metastases that are present but not detectable yet.
In general, the less differentiated and the larger the primary tumor is, the more likely it is to metastasize. However, some large cancers don’t metastasize and some very small ones do. Spreading, or dissemination, strongly influences the prognosis of the patient.
There are three “pathways” cancers can disseminate; either into the body cavity the cancer is in (seeding onto serous membranes), by the lymphatic system (lymphatic spread) or by the blood (haematogenous dissemination). Different types of cancers have ways they “prefer” to spread, so when we find a primary cancer in a specific area can we know where we should look to find metastases. They’re best summed up in tables.
Seeding
| Spread to | Cancers |
| Peritoneum | Ovarian cancer, gastrointestinal stromal tumor, pseudomyxoma peritonei |
| Pleura | Breast cancer, lung cancer |
| Pericardium | Melanoma, breast cancer, lung cancer, squamous cell carcinoma of the head and neck |
Seeding into serous membranes is called carcinosis, like peritoneal carcinosis, pericardial carcinosis or pleural carcinosis.
Lymphatic spreading is more typical of carcinomas; however almost any cancer can spread through the lymphatic system. It’s common for tumors to metastasize into the first filtrating lymph node of the tissue it belongs to. This first lymph node is called the sentinel lymph node, and its importance is this: If the sentinel lymph node is free of metastases can we assume that the primary tumor hasn’t metastasized through the lymphatic system. For example, the sentinel lymph nodes for the testis and ovaries are the paraaortic (or lumbar) lymph nodes and for the breast are the axillary lymph nodes.
However, some cancers spread through the lymphatic but skip the first (sentinel) lymph node, so-called skipping metastasis.
Haematogenous spreading is more typical for sarcomas but can also occur in any cancer. Since all portal blood ends in the liver, and all venous blood ends in the lungs are the liver and the lungs the most frequently involved secondary sites of haematogenous spreading. We can divide haematogenous spreading into three main types, best summed up in a table.
| Spreading through which system | Spreading to | Primary cancers |
| Portal system | Liver | Gastrointestinal cancer |
| Caval system | Lungs | Liver, testis, kidney, thyroid, musculoskeletal system |
| Paravertebral plexus system | Vertebrae | Prostate, thyroid |
Not all metastases can be easily explained by anatomy though. The following examples illustrate that:
- Prostate cancer frequently spreads to bone
- Lung cancer frequently spreads to the adrenals and brain
- Neuroblastomas frequently spread to the liver and bones
- Skeletal muscles, although rich in blood supply, are rarely metastasized into