Table of Contents
Page created on November 2, 2018. Last updated on May 24, 2019 at 17:31
Different organs respond to shock differently. The organ damage depends mostly on the degree of vasoconstriction for the specific organ.
Brain
The brain isn’t really affected by shock, except in the very late phases. There are no morphological or functional changes in the brain during a shock. The body will always try to conserve the blood flow to the brain (and coronaries) in any condition.
Heart
As seen in topic 18 is the coronary circulation auto-regulating. This autoregulation only works until the mean arterial pressure reaches 60-70, below which the circulation will be decreased. This only occurs in late stages of shock. As seen in the same topic will an insufficient coronary circulation affect the endocardium first and epicardium last.
Certain substances like MDF and lysosomal enzymes decrease the myocardial contractility and are present in shock.
Tachycardia develops to compensate for the reduced cardiac output, however tachycardia reduces the diastolic time and therefore the coronary perfusion of the myocardium. This further impairs the contractility, causing a vicious cycle called the Braunwald spiral.
Gastrointestinal tract
The GI-tract and liver receive one third of the cardiac output in rest. All this blood can be redistributed if required.
Because the apexes of the villi are farthest away from the blood supply will they be affected first, while the crypts of Lieberkühn will be affected last. The result is that absorption is impaired long before the secretion. This causes a loss of fluid into the lumen, increased motility and severe, sometimes bloody diarrhoea. Later will the ischaemia affect the muscles and cause hypomotility.
The GI-tract consequences of shock are worse in elderly, who already have reduced GI-tract blood supply due to atherosclerosis. The GI-tract changes can therefore be lethal in elderly while being non-lethal in younger people.
The damaged mucosa can allow bacteria to enter the circulation and complicate the shock.
Pancreas
Ischaemia of the pancreas causes it to release protease enzymes. It also releases a peptide called myocardial depressing factor (MDF), which impairs the myocardial contraction and therefore the cardiac output. This can initiate a vicious cycle where hypoperfusion causes decreased cardiac output which causes more hypoperfusion and so on.
Liver
Shock causes necrosis of the areas around the central veins, which also releases lysosomal enzymes. The detoxifying ability of the liver becomes impaired. The liver’s ability to remove MDF from the circulation diminishes. Liver failure occurs.
Kidneys
The renal blood flow is approximately 20-25% of the resting cardiac output. This flow is physiologically reduced during exercise without problems, as the kidney doesn’t need that much blood. In shock however will the renal blood flow decreases so much that problems occur.
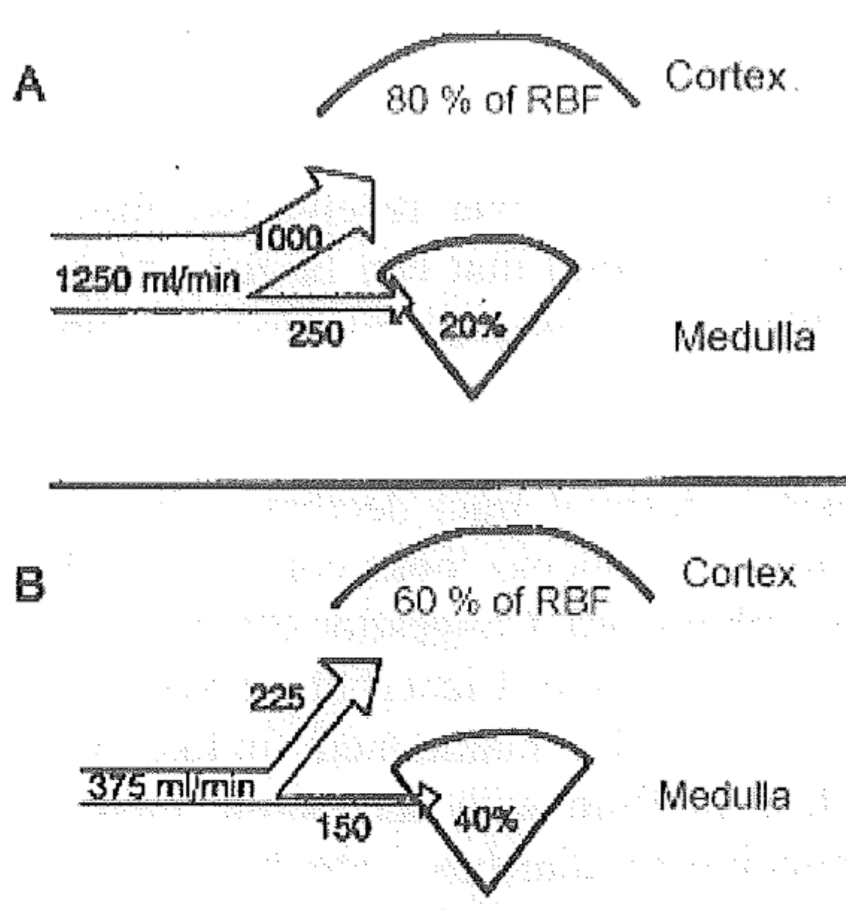
During shock won’t just the renal blood flow and glomerular filtration decrease but there will be hypoxaemic damage of the tubules. This will cause the filtrate to leak back into the circulation, causing oliguria.
Ischaemia of the tubules cause acute tubular nephropathy, a type of acute kidney failure. The glomeruli are usually not affected. The tubules have a much larger energy requirement than the glomeruli. Dialysis helps with the acute tubular nephropathy.
Lungs
Shock causes a condition called acute respiratory distress syndrome or “shock lung”.
Prostaglandins, thromboxanes, leukotrienes, enzymes, free radicals, cytokines and vasoactive substances are transported to the lung via the circulation. These substances damage the endothelium in the pulmonary capillaries. This causes disseminated intravascular coagulation (DIC).
The endothelium damage and inflammation causes capillary permeability to increase. Protein-rich exudate leaks out and accumulates in the interstitium and alveoli, causing pulmonary oedema. The oedema causes breathing to become even shallower, causing further hypoxaemia. Phagocytes migrate into the lung tissue and damage interstitial cells, collagen and elastic fibres.
Pituitary
The portal circulation of the pituitary is especially sensitive to DIC. The result is hypoxic damage, resulting in permanent pituitary insufficiency. Hypovolaemic shock (and possibly other shocks as well) can lead to Sheehan syndrome, where the pituitary gland is destroyed and can’t produce hormones.
Adipose tissue
Adipose tissue receives basically no circulation in shock. The dying adipocytes release lactic acid and lipase. This worsens the DIC. Because of this is the prognosis of shock much worse in obese than in normal-weight patients.
Hi Doctor Nikolas ;),
A question came up my mind, you say that shocks get complicated in obese people as they have a lot of adipose tissues, so there will be loads off lactic acid in the vessels. In case of a distributive shock would obese people have no “ cold phase “ as they have alot of lactic acid that can cause Vasodilation, or their “cold phase” will be reached when they are in a severe severe case ?
Thank you !
Good question. I don’t know (but it surely isn’t important to know for the exam).