Page created on December 14, 2018. Last updated on May 23, 2020 at 13:46
The tubules work hard
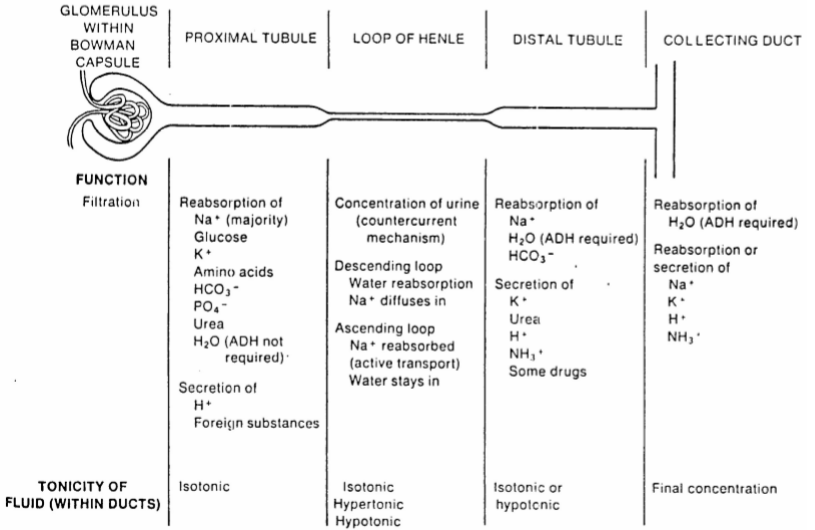
Despite there being 180 L of ultrafiltrate produced each day is the urine output only 1 – 1.5 L/day, indicating that the tubules must do a lot of reabsorption. The urine also has a very different composition from the ultrafiltrate. When the tubules dysfunction will both the quantity and the quality of the urine be different.
This is a good time to introduce some new (or not) terms. Specific gravity is a measure of how concentrated the urine is compared to pure water. It’s calculated like this:
![]()
From this formula can we understand that if the concentration of your urine is the same as the concentration of pure water would the specific gravity be 1. However, because there are always some molecules or ion in urine (urine cannot be pure water) will the concentration of urine always be higher than the concentration of water, so the number will always be above 1. If your urine was just pure water would the specific gravity be 1.000. As we said, that’s impossible. The absolute lowest specific gravity of urine possible is 1.001, meaning that the urine is just barely more concentrated than pure water (just 0.1% more concentrated than water).
The normal value for specific gravity of urine is 1.010 – 1.012 if a person is well hydrated, but in pathological conditions or overhydration it can be as low as 1.001 (but never lower!). In exsiccosis the specific gravity can be around 1.030 – 1.035. There is no upper roof for specific gravity, as it can be above 1.035 in e.g. diabetes mellitus.
Hyposthenuria is the condition where the kidney loses its ability to concentrate or dilute the urine. This usually decreases the specific gravity of the urine, but we will get back to that later.
Primary tubular dysfunctions
We have both congenital and acquired primary tubulopathies. They are primary in the sense that there is no other renal damage in the background (at least in the early phase).
Renal tubular acidosis (RTA) is a condition where the tubules can’t excrete H+, so the kidney can’t acidify the urine, causing acidosis. It can have both congenital and acquired causes. There are three (important) types.
- RTA type 1 where the H+ excretion in the distal tubules is defective
- RTA type 2 where the bicarbonate reabsorption in the proximal tubules is decreased
- RTA type 4 where there is hypoaldosteronism or tubular cells are resistant to aldosterone. Hyperkalaemia also occurs.
See topic 76 for more details on RTA I and II.
It’s still uncertain if what was originally considered RTA type 3 actually exists, which is why the types are so weird. We’ll come back to this in the acidosis topics.
Renal diabetes insipidus can also be either congenital or acquired. It occurs when the distal and collecting tubule cells are AVP resistant, making them unable to concentrate the urine. A lot of water is lost in the urine.
Tubular hypoxia, whether due to hypoxaemia, ischaemia or transplant rejection will cause non-specific tubular damage with hyposthenuric polyuria.
Some toxins and drugs can cause tubulointerstitial nephritis, like mushroom toxins, antifreeze, drug effects and hypersensitivity reactions. SNGFR will increase in the unaffected nephrons, damaging them as well.
Certain metabolic disorders, like uric acid, oxalate, hypokalaemia and hypercalcaemia can damage the tubules. Hypercalcaemia can cause metastatic calcification of the tubules, and can occur from paraneoplastic syndromes.
Autoimmune diseases like SLE or Sjögren syndrome can damage the tubules.
Fibrosis can compress and damage the tubules.
Renal glucosuria is a congenital condition where the person has limited ability to reabsorb glucose in the proximal tubule. This may cause osmotic diuresis.
Renal aminoaciduria is another congenital condition where the amino acid reabsorption capacity is reduced. It usually causes cysteine stones and amino acid deficiencies.
Renal phosphaturia may develop if there is hyperparathyroidism or if the tubules are too sensitive to PTH. This may cause rickets (bone weakness).
Fanconi syndrome is a very rare congenital or acquired disorder characterised by glucosuria, aminoaciduria, phosphaturia and bicarbonate loss.
Secondary tubular dysfunctions
The glomerular filtration rate (GFR) is the sum of the SNGFR for all functional tubules in both kidneys. It’s therefore a measure of the total glomerular filtration rate of both kidneys.
These dysfunctions occur because of a primary problem with filtration, which causes the tubules to have to adapt and compensate for the filtration problem.
If SNGFR is decreased the filtrate will flow more slowly through the tubules, allowing the tubules more time to reabsorb salts and fluid. This causes more fluid to be reabsorbed into the plasma and less urine to be produced, which normalizes the plasma volume despite the reduced filtration.
In cases where the number of functioning nephrons decreases the SNGFR of the remaining functional nephrons increases to compensate. This allows the kidneys to maintain the GFR for some time. However, the SNGFR can only increase to a certain point, so a point will be eventually be reached, where further loss of nephrons causes the GFR to decrease as well.
If the SNGFR is elevated and the tubules reabsorbed the same amount of fluid and salt as they do when the SNGFR is normal, a lot of water and fluid would be excreted and lost. So, the tubules must also be increase the reabsorption of certain salts and water to avoid losing them.
However, the SNGFR increases because the kidneys wants to get rid of more salt and fluid. If the tubules increased their reabsorption so much that all excess filtrate would be reabsorbed, the kidney wouldn’t get rid of more salt and fluid. It would just reabsorb the excess filtered salt and fluid.
Therefore, when the SNGFR increases, some, but not all, of the excess filtrate should be reabsorbed. The tubules must balance their reabsorption and secretion to ensure homeostasis. The process where the tubules adjust their degree of reabsorption and secretion in response to increased SNGFR is called tubular adaptation or the magnification phenomenon.
The tubular adaptation is different depending on the ion in question. The adaptation is very good for Na+ and K+, meaning that when the GFR decreases the kidney can maintain normal levels of sodium and potassium very well. This prevents hypernatraemia and hyperkalaemia. For sodium this is accomplished by increased level of natriuretic substances, which ensure that the reabsorption of sodium is not increased too much. For potassium this is accomplished by increased level of aldosterone, which increases potassium secretion. The adaptation of these two ions is so good that the plasma concentration of these salts doesn’t increase until the GFR is almost 0.
When the SNGFR increases the tubular excretion of H+ and phosphate increases, preventing metabolic acidosis and hyperphosphataemia. For phosphate this is due to increased PTH levels, which decreases phosphate reabsorption. The adaptation of H+ and phosphate prevents the plasma concentration of these ions to increase until the GFR reaches 25% of normal.
The tubular adaptation of urea and creatinine is poor. There are no substances like natriuretic peptides, aldosterone or PTH to adapt the tubular reabsorption and secretion for nitrogen-containing waste products like urea and creatinine. The result is that urea and creatinine will start to accumulate in the blood as soon as the GFR reaches 40 – 50% of normal.
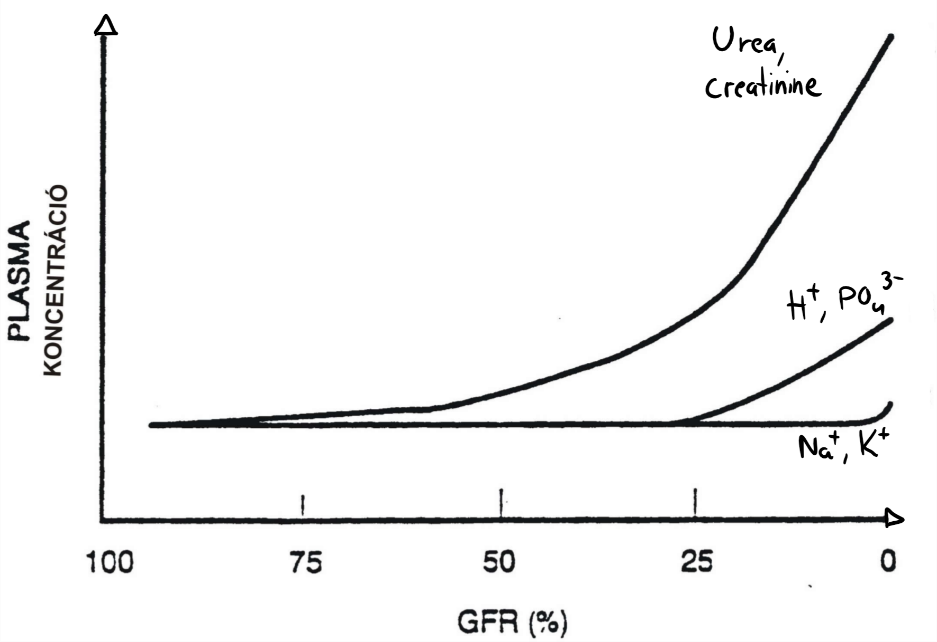
This graph shows the plasma concentration of different substances in correlation with the GFR. From this figure we can understand that hypernatraemia and hyperkalaemia only occurs when GFR is almost zero, while azotaemia (urea and creatinine in blood) occurs already when GFR drops to half its normal value.
hello greek.doctor.
in the paragraph of some toxin and drugs :
should,nt be interstitial nephritis ?
since tubulointerstitial nephritis is caused by bacteria and anything else that damages tubules other than bacteria is called interstitial nephritis?
TIN is a general term used to refer to inflammatory kidney diseases that involve the interstitium and tubules.
Pyelonephritis is the term we use when bacteria is involved because the renal pelvis is prominently involved, and interstitial nephritis is generally reserved for cases of TIN that is nonbacterial in origin.
Both Pyelonephritis and interstitial nephritis are TIN, since its a general term.
hope this helps
Cheers
thanks.
Hello!
One thing i do not understand, when we have decreased RBF say due to hypovolemia, the kidneys try to increase Na+ and water retention? i dont quite understand the last part with magnification, when GFR decreases, then Na excretion incresases, isnt that kind of counterproductive in regard to the hypovolemia?
When the RBF decreases the SNGFR decreases. The magnification phenomenon is about when the SNGFR increases, due to nephron loss in chronic renal failure.