Table of Contents
Page created on April 2, 2019. Last updated on June 14, 2022 at 12:26
Introduction
Hormones are endogenous messenger molecules that are produced in glands or single cells. Hormones can act by endocrine, neurocrine, paracrine or autocrine pathways. The endocrine hormones are those which will be examined in the next bulk of topics.
All hormones act by binding to special receptors on their target tissues or organs. Hormone release may be continuous or pulsatile or periodic and their effect may be altered based on this. For example will continuous release of GnRH suppress LH and FSH, but pulsatile release of GnRH stimulates LH and FSH production.
The effect of a hormone can be insufficient despite the levels being normal, if the receptors aren’t working correctly. The receptors themselves may be defective or down-regulated, like in insulin resistance and renal diabetes insipidus, or the receptors may be blocked by antibodies or false transmitters.
The action of the hormones on a cell also depends on the state of the cell. In histotoxic hypoxia will the hormone-response of the cells be low despite normal function of the hormones themselves and their receptors.
Hormone receptors are usually specific for a given hormone, but not always. For example can cortisol activate mineralocorticoid receptors too. This is called “spillover”.
Persistent high levels of a hormone usually cause the hormone receptor to be downregulated as a “defence” against a large hormone response.
We can classify the different hormones according to their structure:
- Peptide hormones
- Insulin
- Glucagon
- Secretin
- Parathyroid hormone
- Steroid hormones
- Aldosterone
- Cortisol
- Sex steroids
- Amine hormones
- Catecholamines
- Thyroid hormones
- Glycoproteins
- FSH
- LH
- TSH
Peptide hormones are produced by enzymatic cleavage of prohormones. They are stored inside the cells that secrete them partly in prohormone form and partly in the “mature” form. These hormones then get into the circulation by exocytosis. These hormones act on membrane-bound receptors.
Steroid hormones are synthesized from cholesterol and are stored in only small amounts in the producing cells. They are secreted by passive diffusion. Since these hormones are lipid soluble will they cross the cell membrane of the target cell by diffusion, where they bind to receptor in the cytoplasm. The hormone-receptor complex enters the nucleus and acts on the transcription there.
Amine hormones like the catecholamines act similarly to the peptide hormones, while thyroid hormones act similarly to the steroid hormones. These hormones are stored before secretion.
Glycoproteins act like the peptide hormones. These hormones are also stored before secretion.
Most endocrine organs are controlled by the hypothalamus and pituitary. This forms a hypothalamo-pituitary axis, which you can see on the figure below.
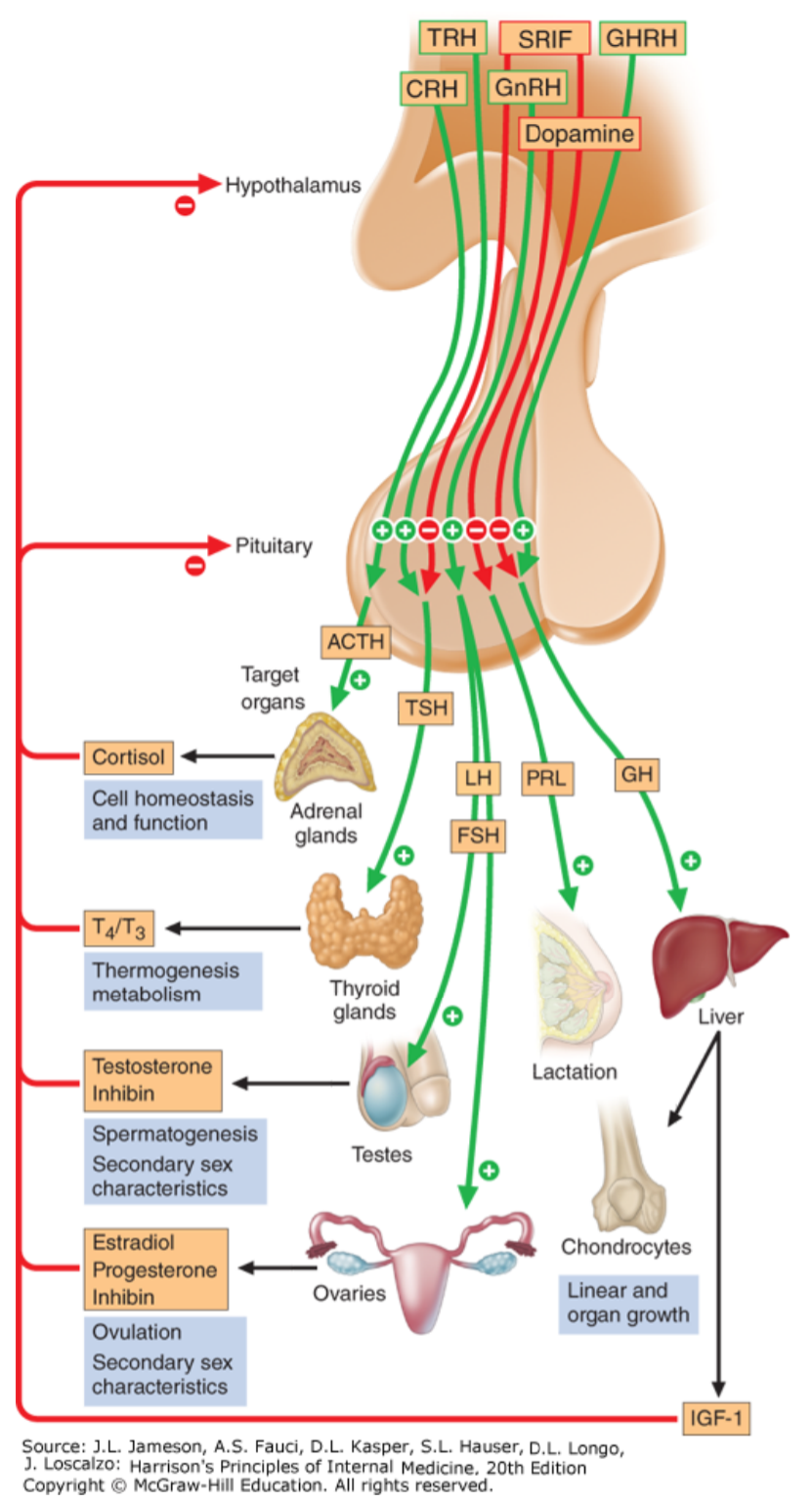
Endocrine dysfunction
The function of an endocrine organ can be either increased or decreased, both of which can cause diseases or symptoms.
Endocrine hypofunction, the decreased effect of an endocrine organ, can occur either if there is something wrong with the organs that produce the endocrine hormone or if there is something wrong with the organs the hormone acts on. Here are some of the factors that can lead to endocrine hypofunction:
- Decreased production of the hormone
- Production of an abnormal variant of the hormone
- Disorder of the hormone receptors on the target tissue
- Binding of antagonists to the receptors
- Disorder of the second messengers of the hormone receptors
- General disorders of the target cells, like hypoxia
We distinguish three types of endocrine hypofunction, based on where the problem lies:
| Organs in the hypothalamic-pituitary axis | Organs not in the hypothalamic-pituitary axis | |
| Primary hypofunction |
Hypofunction of the peripheral gland |
|
| Secondary hypofunction | Hypofunction of the pituitary | Decreased stimulation of the peripheral gland |
| Tertiary hypofunction | Hypofunction of the hypothalamus |
– |
The classification should be thought of like this: For thyroid hormone, which is controlled by the hypothalamic-pituitary axis, primary hypothyroidism would mean a defect in the thyroid gland itself, while secondary hypothyroidism means a defect in the TSH production of the pituitary gland, which leads to hypofunction of the thyroid gland.
For parathyroid hormone (PTH), which isn’t controlled by the hypothalamic-pituitary axis, primary hypoparathyroidism would mean a defect in the parathyroid glands themselves, while secondary hypoparathyroidism would mean low production of PTH in response to hypercalcaemia.
Endocrine hyperfunction, the increased effect of the endocrine organ may also occur either if there is something wrong with the organs that produce the endocrine hormone or if there is something wrong with the organs the hormone acts on, or due to the spillover effect. Here are some factors that can lead to endocrine hyperfunction:
- Increased production of the hormone
- Increased sensitivity of the receptors
- Spill over by other hormones
The classification of endocrine hyperfunction is similar to that of hypofunction:
| Organs in the hypothalamic-pituitary axis | Organs not in the hypothalamic-pituitary axis | |
| Primary hyperfunction |
Hyperfunction of the peripheral gland |
|
| Secondary hyperfunction | Hyperfunction of the pituitary | Increased stimulation of the peripheral gland |
| Tertiary hyperfunction | Hyperfunction of the hypothalamus |
– |
For thyroid hormone, which is controlled by the hypothalamic-pituitary axis, primary hyperthyroidism would mean a problem with the thyroid, causing it to produce too much thyroid hormones, while secondary hyperthyroidism means that the pituitary produces too much TSH.
For aldosterone, which isn’t controlled by the hypothalamic-pituitary axis, primary hyperaldosteronism would mean there is something wrong with the adrenal cortex that causes it to produce too much aldosterone, while secondary hyperaldosteronism means that the stimulus that stimulates aldosterone production is excessive (the RAAS).
Hypopituitarism
Hypopituitarism refers to the condition where there is inadequate production of one or more anterior pituitary hormones as a result of damage to the pituitary or hypothalamus. If production of all pituitary hormones is impaired is the condition called panhypopituitarism.
Hypopituitarism will cause downstream hypofunction of the endocrine organ whose trophic hormone has its production impaired. For example will deficiency of TSH cause hypothyroidism.
What’s interesting in hypopituitarism is that the different pituitary hormones are usually lost in a special sequence. When the pituitary is damaged, the order of hormones lost is as follows: GH -> FSH and LH -> TSH -> ACTH -> prolactin.
There are many causes of hypopituitarism. The most common cause is the compression of the pituitary due to a tumor, but here are some other important causes:
- DIC (Sheehan syndrome)
- Pituitary apoplexy
- Cranial trauma – especially if it affects the base of the skull
- Irradiation
- Granulomatous disease – like sarcoidosis or tuberculosis
- Infiltrative disease – like amyloidosis or haemochromatosis
The symptoms depend on which hormones are affected, the gender and the age of the patient:
| Lost hormone (in the order they are lost) | Symptoms in children | Symptoms in adults |
| GH | Dwarfism | Many (see topic 57) |
| FSH, LH | Delayed puberty | Anovulation, amenorrhea, loss of libido, impotence |
| Prolactin |
– |
No production of milk after delivery |
| TSH | Hypothyroidism: Cold intolerance, weight gain, lethargy, constipation, dry skin | |
| ACTH | Weakness, hypotension, hyponatraemia, hypoglycaemia | |
There is no severe mineralocorticoid deficiency as this is regulated mainly by RAAS and not by ACTH. Thus, panhypopituitarism can be survived.
Sheehan syndrome is a condition where the pituitary gland is damaged due to shock and DIC during delivery. During pregnancy there will be hypertrophy of the prolactin-producing regions of the pituitary, which makes it sensitive to ischaemia. When a lot of blood is lost because of complications with the delivery will the resulting hypovolaemia be enough to cause ischaemia and necrosis of the pituitary. Mostly the production of prolactin, FSH and LH are affected. The absence of prolactin means that the mother doesn’t produce milk, which is often the first symptom. Absence of FSH and LH causes anovulation later.
Pituitary dwarfism is described in topic 57.
Hypothalamic disease
Unfortunately, this condition isn’t called hypohypothalamusism.
Hypothalamic disease refers to a disorder that primarily affects the hypothalamus. It’s a rare condition that is usually only seen in surgery, traumatic brain injury, tumors and radiation. It causes symptoms such as:
- Disorders of appetite, food intake and body weight regulation
- Disorders of thermoregulation
- Disorders of sleep
- Disorders of memory
- Disorders of thirst
- Central diabetes insipidus
The posterior pituitary and it’s disorders
The posterior pituitary contains axon endings of hypothalamic neurons. Oxytocin and vasopressin (AVP or ADH) are released from these axons by neurosecretion. Disorders of the posterior pituitary are mostly caused by brain tumors, traumatic brain injury or neurosurgery.
Oxytocin is involved in lactation (but not milk production itself) and in uterine contractions during labour.
Vasopressin is both an antidiuretic and a vasoconstrictor. AVP acts on AVP receptors in the collecting ducts and moves aquaporin channels to the membrane surface, which allows reabsorption of water (but not salt!). AVP is produced in response to increased plasma osmolarity.
Central diabetes insipidus is the condition where the AVP production is impaired. This causes primary polyuria and inability to concentrate the urine. It should be distinguished from renal diabetes insipidus, where the AVP receptors in the kidney are defective, but the symptoms are similar.
Syndrome of inappropriate ADH (SIADH) is a condition where the level of AVP is abnormally elevated. It may be due to the pituitary secreting too much of it, or if there is an ectopic source of AVP. The latter is especially characteristic as a paraneoplastic syndrome of small cell lung carcinoma. It’s usually asymptomatic but the resulting hyponatraemia can be significant and cause seizures or altered consciousness.
You think I can use the pathology topic for this rather?
You can use whatever you like. I haven’t compared them to see if the pathophysiology topic contains anything in particular which the patho topic lacks.
Isn’t prolactin the last pituitary hormone that is lost in case of hypothyroidism
Think so, corrected now.