Page created on October 22, 2018. Last updated on May 21, 2019 at 17:39
For respiration to be efficient must all these processes work correctly:
- Ventilation
- Diffusion of gases across the alveolo-capillary membrane
- Transport of gases in the blood
- Oxygen uptake of cells
We’ll start by examining the regulation of these processes.
Regulation of respiration
Neurons that control the breathing muscles sit in the respiratory centre in the medulla oblongata and pons. The respiratory centre receives inputs from chemoreceptors, mechanoreceptors, cerebral cortex and the hypothalamus.
- Chemoreceptors measure the pH, pCO2 and pO2 in the blood
- Mechanoreceptors receive inputs from the respiratory muscles and other skeletal muscles
- The cerebral cortex gives conscious control of respiration
- The hypothalamus controls respiration during stress and anxiety
The most important are the pCO2 and H+-sensitive chemoreceptors. The pO2 receptors are only of secondary importance. In physiological conditions is it the pCO2 level that controls ventilation. Only when there is a hypocapnia will pO2 start to influence breathing.
When the pCO2 level has been high for a long time, like in chronic hypercapnia in case of COPD will the CO2-sensitivity of the chemoreceptors be reduced. This means that the respiratory won’t react correctly to a hypercapnia and breathing might be driven by pO2 instead. If the patient then receives oxygen therapy will their pO2 become normal, so there is nothing left to drive the respiratory centre! The patient will therefore slow down their breathing, so a severe hypercapnia develops. This can cause life-threatening narcosis or coma, so-called CO2-narcosis.
Abnormal breathing patterns
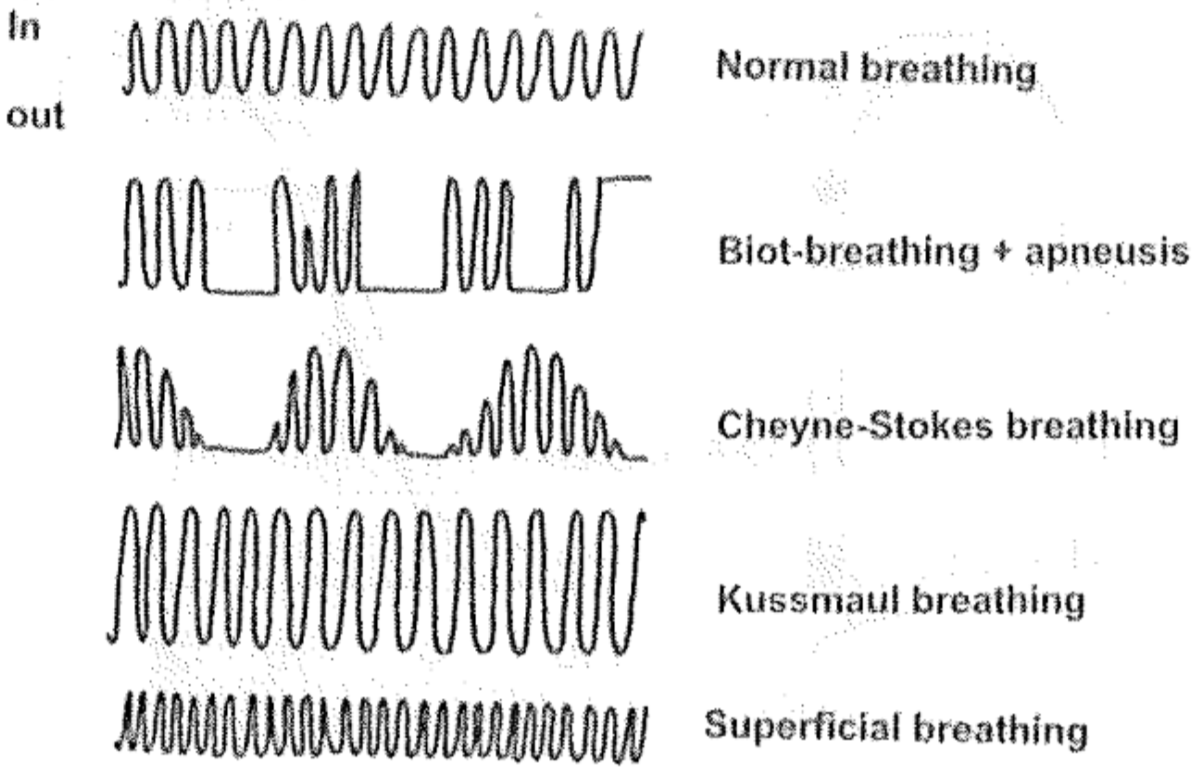
Even if respiration is continuous it may be abnormal. Rhythm, frequency and volume of breathing must all be closely regulated, and abnormal regulation may lead to abnormal alveolar ventilation. Hypopnoea means that the tidal volume is smaller than needed, meaning that the breaths are too shallow. Hyperpnoea means the opposite. In apnoea will respiration stop after expiration, meaning that there will be no inspiration. Apneusis is when respiration stops after an inspiration (so there is no expiration).
A breathing pattern called Cheyne-Stokes breathing is characterized by an abnormal breathing pattern that goes like this: first is the size of breathing large, then becomes smaller until breathing stops completely. Breathing starts again by itself, and the cycle repeats. In other words what happens is hyperpnoea – hypopnoea – apnoea – hyperpnoea – hypopnoea – apnoea and so on. The pattern is also called crescendo-decrescendo with apnoea following. The periods of apnoea can be up to 20 seconds long.
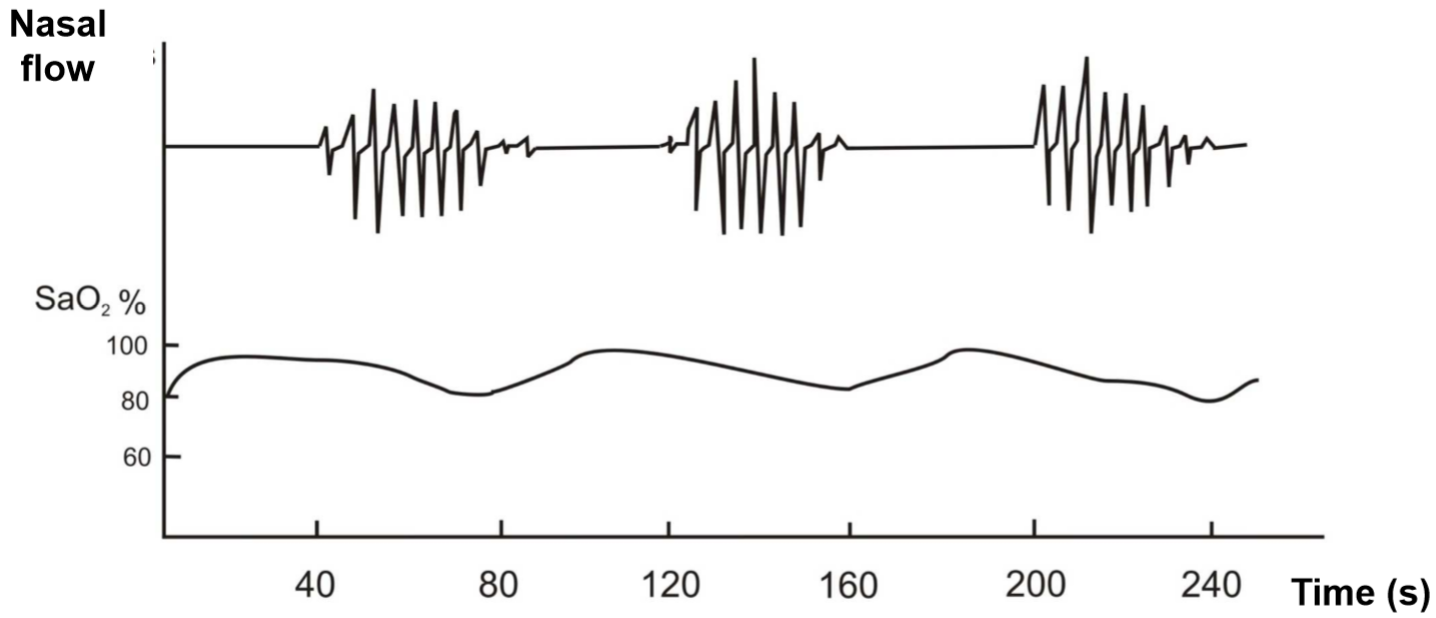
Cheyne-Stokes breathing is seen in sleeping neonates as their respiratory system isn’t quite finished yet. It can be seen in adults in high altitudes and in patients with congestive heart failure as well.
In Biot-breathing both the frequency and the depth of breaths are chaotic. Periods of apneusis may occur. It’s often seen in premortal patients.
Kussmaul breathing is characterized by hyperpnoea. It occurs when blood pH is decreased, often because of ketoacidosis associated with diabetes. Hyperpnoea is induced to try to get rid of the excess CO2 to increase pH.
Sleep apnoea syndrome (SAS)
Breathing patters change slightly during sleep even in healthy people, however without significant hypoxia or hypercapnia. A patient with SAS has over 30 periods of apnoea and hypopnoea that lasts more than 10 seconds every night. This causes significant changes in blood gas levels. Two types exist: central and obstructive sleep apnoea.
Central sleep apnoea is idiopathic and the most severe of the two forms. It’s caused by defective autonomic regulation of breathing and is present from birth. It can lead to insufficient or absent breathing during sleep. The worst form is the so-called curse of Ondine, in which there is no spontaneous breathing during sleeps. In other forms will the defective autonomic breathing regulation only become significant in old age. The apnoeic periods cause significant hypoxia and hypercapnia. In central sleep apnoea there is no movement of the chest during the apnoeic periods, unlike the other type.
Obstructive sleep apnoea is the most common form of sleep apnoea. It’s caused by complete or partial obstructions of the upper airways. In healthy people the muscle tone of the pharynx and soft palate will decrease in the deepest phases of sleep. In people with obstructive sleep apnoea will this decreased muscle tone cause the muscles to be so loose that they can obstruct the air flow. The chest will still expand but no air will enter. The air flow can stop completely, which will trigger an arousal in the CNS. The arousal triggers sympathetic activity and catecholamine release, which causes the person to almost wake up.
Obstructive sleep apnoea-suffering patients have lower quality of sleep because the deep phases of sleep are constantly interrupted by these arousals during the night. The consequence can be daytime sleepiness and fatigue. The decreased air flow causes a global respiratory failure (pO2 < 60 mmHg, pCO2 > 50 mmHg).
The catecholamine surge during the apnoeic periods isn’t good either. It causes tachycardia, which increases oxygen need of the heart. In people with coronary stenosis due to atherosclerosis or with stable angina can the increased oxygen need trigger an infarct or angina attack.
Both types of sleep apnoea syndrome are more frequent in males and gets more pronounced with increasing age and body weight.
Hi 🙂
I have a question regarding the mechanism of Kussmaul breathing. How does it increase pH by removing CO2? I thought CO2 could bind H+ and form HCO3-, thereby increasing blood pH (by removing H+ from the blood)
CO2 spontaneously combines with water to form H2CO3, which is a diprotic acid. By removing CO2 you decrease the amount of H2CO3 produced.
CO2 can’t bind H+ and form HCO3-. Where would the extra oxygen atom come from?
Ahh, that makes sense. Thank you!!